Interview with Gwenola Le Dréan – Part 1

Today we have the pleasure of interviewing Gwenola Le Dréan, physiologist and specialist in the gut-brain axis, who is a researcher at INRAE (the French National Research Institute for Agriculture, Food and the Environment) at the UMR 1280 “Physiopathology of Nutritional Adaptations” laboratory, which she co-directs in Nantes.
Your research aims to understand how unbalanced perinatal nutrition increases the risk of metabolic and cognitive diseases later in life. Among other things, you are studying the communication between hormone-producing intestinal cells and sensory neurons that signal the presence of nutrients and bacterial metabolites in the intestinal lumen to the brain.
First of all, can you tell us about the concept of the developmental origins of health and disease?
The concept of the developmental origins of health and disease, known by the acronym DOHaD, was first proposed in the 1990s by an English doctor, D.J. Barker. He highlighted that an altered nutritional environment during the prenatal period predisposes individuals to cardio-metabolic diseases later in life, such as type 2 diabetes, hypertension and obesity. Barker’s epidemiological studies are based on monitoring individuals whose birth weight and weight at one year of age were recorded in the Hertfordshire cohort, as well as those who were exposed to famine in utero during the “Dutch Hunger Winter” in 1944. These studies led him to conclude that the metabolism and growth of a foetus in a situation of nutritional deficiency adapts to this impoverished environment. This metabolic adaptation induces a “thrifty phenotype” that protects the development of key organs such as the brain and kidneys, at the expense of growth.
However, when faced with the abundance of calories frequently found in Western diets high in fat and sugar, this phenotype is not always able to adapt and contributes to the development of chronic metabolic diseases, such as cardiovascular, metabolic and cognitive disorders later in life.
Interestingly, it has since been shown that an unbalanced perinatal nutritional environment – whether it is deficient or excessive in calories or essential nutrients – creates a similar possibility of developing these diseases, due to the high plasticity of developing organs.
While this perinatal window of the first 1,000 days, from conception to the first two years of life, is extremely sensitive to the environment, it also provides an opportunity for prevention and intervention to correct or limit the risk of future diseases.
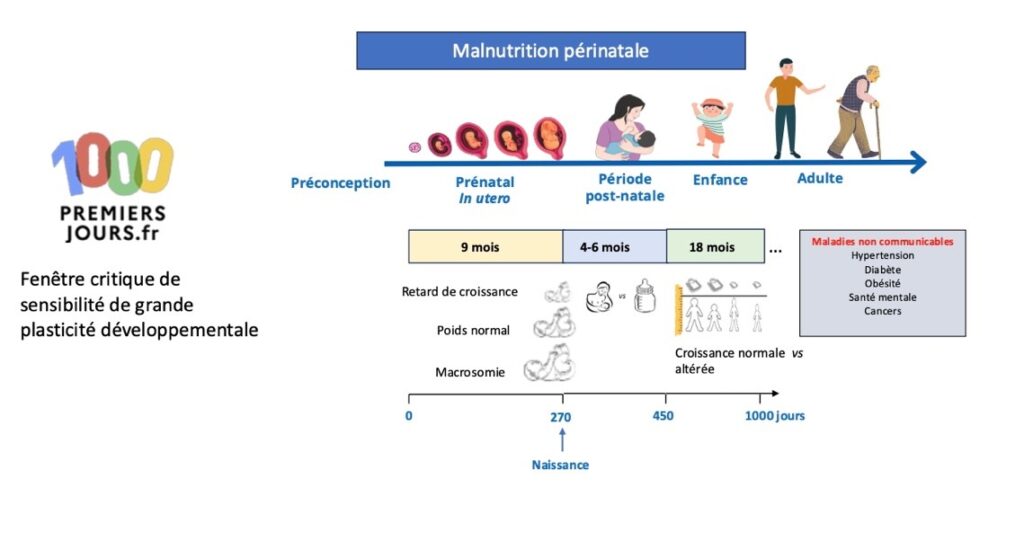
The concept of DOHaD illustrated by the consequences of perinatal malnutrition on the risk of chronic diseases later in life. Created by BioRender and adapted from Mayneris-Perxachs and Swann, 2019.
Thank you. And to better understand these issues, can you explain the role of nutrition in the first 1,000 days on the microbiota-gut-brain axis?
It is important to note that the gut-brain axis initially defines the bidirectional communication that takes place between these two organs. This communication has been known and described for several decades in the regulation of food intake. When food arrives, the gut sends nutritional and digestive signals – such as distension of the stomach and small intestine, production of hormones that regulate food intake, etc. to the brain stem, which sends back signals regulating digestive functions such as the motility of the intestinal wall muscles, which ensure the transit of food. All this communication is carried out via the bloodstream but also via the vagus nerve, which integrates mechanical and hormonal signals as well as those from the nervous and immune systems of the digestive tract. Thus, beyond the establishment of satiety or the state of not being hungry, regulated reflexively through this communication, the signals transmitted by the gut-brain axis play a central role. This role is decisive not only in regulating the energy balance between calorie intake and expenditure, but also in higher cognitive functions such as learning, memory, and the perception of visceral pain, stress and anxiety.
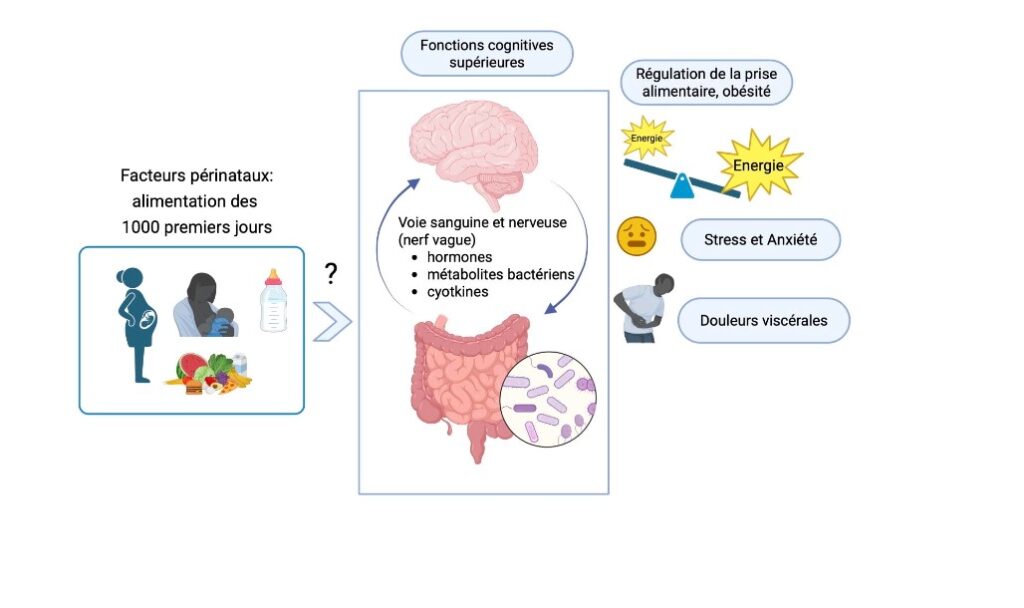
What role does nutrition during the first 1,000 days play in the microbiota-gut-brain axis and its involvement in certain future health disorders? Created by BioRender.
Can you also explain what the microbiota does?
More recently, a new player has entered the picture in our understanding of this axis: the microbiota and its 10 trillion organisms, which are mainly, but not exclusively, bacteria. The term “commensal flora” was sometimes used to define these microorganisms with which we share our nutrients. This microbial ecosystem is now called the “microbiome” by specialists in the field, but the term microbiota remains very common.
We host our microbiota in a living space that is conducive to its growth, the gut, and we feed it. In return, the microbiota uses a number of molecules that our digestive tract cannot assimilate, produces molecules that are important for our body, such as vitamins, and contributes to the development of the intestines and the immune system. It also helps prevent infections by producing antimicrobial agents while occupying the space that certain pathogenic microorganisms could take up in our intestines. Finally, the gut microbiota also sends messages to our brain by producing molecules such as hormones that regulate appetite and neurotransmitters that ensure communication between neurons.
Animal models raised in sterile conditions – i.e. without microbiota – or treated with antibiotics remain essential for understanding the role of microbiota in the development of the immune system, the brain and behaviour. It should also be noted that dysfunction of the microbiota-gut-brain axis is often reported in the pathophysiology of many diseases, including mental illness, neurodegenerative diseases, obesity and inflammatory bowel disease.
This axis therefore represents a prime target for developing new therapeutic approaches! Of course, experimental research must nevertheless be transposable to humans. This is why translational research aimed at demonstrating the causal role of the microbiota in these disorders is currently underway.
And why are the first 1,000 days so crucial to the function of the microbiota?
The first 1,000 days are truly critical windows of development. It is during these first 1,000 days that the gut microbiome, and the metabolites it produces, interact with the infant’s immune system and central nervous system. Bacterial metabolites produced by the maternal microbiota can affect neurodevelopment during the foetal period via the placenta, as is the case with nutrients. However, the baby’s neurodevelopment continues in the postnatal period as the microbiota colonises the digestive tract. Newborns are born sterile. At the moment of delivery, it encounters its mother’s vaginal and faecal microbiota if it is born vaginally, or, if it is born by caesarean section, the skin microbiota and that of the delivery room! This neonatal microbiota then settles like a pioneer on virgin territory, proliferating and colonising the digestive tract. This colonisation becomes more complex depending on the child’s diet and environment until the age of 2, reaching a relatively stable composition – although still debated – around the age of 4-6.
We know that diet is a major factor in the variation of microbiota composition in adults. It is therefore entirely conceivable that a maternal microbiota modified by an unbalanced diet, transmitted at the time of birth, could affect the future health of the baby. In a rodent model, we were able to demonstrate in the laboratory that inocula prepared from the microbiota of obese or thin mothers, distributed to their young at birth and in the days that followed, had different effects on their eating behaviour throughout their lives. These inocula are vaginal, faecal and milk-derived microbiota. It appears that the pups that received microbiota from obese mothers showed changes in their eating behaviour, tending towards hyperphagia. Interestingly, in this model, the composition of the microbiota transmitted at birth did not persist over time. It certainly contributed to the assembly of the final microbiome, but it was not the adult microbiome that explained the differences in eating behaviour observed. Thus, it appears that it was indeed the microbiota received at birth that was responsible for the differences, and we can therefore assume that this neonatal microbiota had an effect on neurodevelopment in early life, with consequences for eating behaviour in adulthood. Other studies of standard microbiota transfer from conventional mice to mice raised without intestinal microbiota – axenic mice – have shown that the microbiota received at a young age has a greater effect on exploratory behaviour and stress response than that received in adulthood.
The postnatal window is therefore a crucial period for the assembly and construction of the baby’s gut microbiome. The baby will receive its mother’s microbiota as it has been shaped by maternal nutrition, including her eating habits and behaviours.
End of the first part of this interview. To access to the 2nd part of this interview, click here


